More on Degrees of Freedom
When faced with the task of devising a control scheme for a process it
is necessary to know how many of the process variables am I entitled
to attempt to regulate. By process variables we mean temperatures,
pressures, compositions, flowrates or component flowrates. The answer
arrived at is known as the number of degrees of freedom.
What follows is a summary of a method for analysis of the number of degrees of freedom which is described in:
Ponton JW, 1994, Degrees of Freedom Analysis in Process Control,
Chemical Engineering Science, Vol. 49, No. 13, pp 1089 -
1095.
In fact it is possible to understand the key concepts from a much simpler
viewpoint. This is described here.
Definitions
Before we start analysing the degrees of freedom it is perhaps wise to
define a few terms.
- Degrees of Freedom of a process
- The number of process variables which can be set by the designer,
operator or control system.
- Control Degrees of Freedom
-
- The number of the above types of process variable which may be set
once non-adjustable design variables have been fixed.
- The number of manipulated variables which may be used in control loops.
- The number of SISO control loops.
- The number of regulated variables in the control loops.
The procedure which will be described is a counting process which
identifies potential manipulations, but does not directly identify
variables which may be regulated.
Derivation
The technique can be derived by applying the Kwauk method. This was
first developed by Kwauk [1952] and later by Smith [1963]. The equation
to be solved is
Degrees of Freedom = Unknowns - Equations
Unknowns
There are two types of streams - process streams and energy streams.
If we take a process stream containing C components then the unknowns
are C flowrates, temperature and pressure. An energy stream has 1
unknown associated with it.
Equations
The equations are best thought of as types of equations i.e.
component balances, energy balances, equilibrium equations, equivalent T
and P etc.
Degrees of Freedom
Once the final number of D.O.F. have been found then it is necessary to
decide which of these are already fixed and which can be controlled.
Example 1 - Simple Blender
Let us take, for our first example, a simple blending process. Assume
that the mixing takes place in the vapour phase. There two inputs and
one output as shown in the figure below.
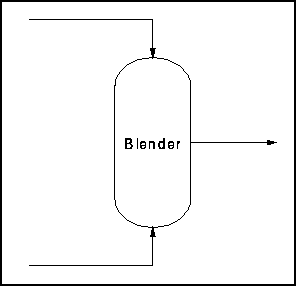
Figure 1 - Simple Blender
It is possible to apply the above equation to evaluate the number of
degrees of freedom
Unknowns
3 streams with C+2 unknowns = 3C + 6 Unknowns
Equations
- C Mass Balance Equations
- 1 Energy Balance Equation
- C+1 Equations
Degrees of Freedom
D.O.F = 2C + 5
However in this system we know the composition, temperature and pressure
of the two input streams. This adds on a further
2C + 2 Constraints
Hence there are 3 D.O.F and so we can fix the output composition,
pressure and throughput.
A typical control scheme is shown below.
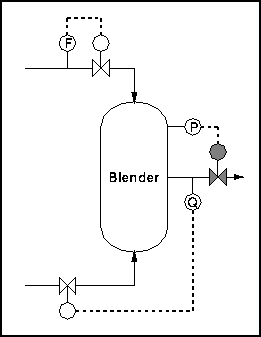
Figure 2 - Simple Blender with Control
The question now is Is there a relationship between the control degrees
of freedom, the number of streams and the number of phases present.
In the paper it is proved theoretically that there is such a
relationship.
It is possible to
- Number all the streams associated with the unit
- Count all the phases
- Determine the number of extra phases ie the (number of phases
- 1)
- This number is also the number of interfaces
- Subtract interface count from stream count giving answer
Hence for the example discussed earlier
Blender
- No. of Streams = 3
- No. of Extra Phases (interfaces) = 0
- CDF = 3 - 0 = 3 - as before
This equation is extremely simple to use and discards the need to think
about unknowns and equations.
The next example we will look at is the adiabatic flash. Note that we
will be returning to this example in a later section. A diagram is
shown below.
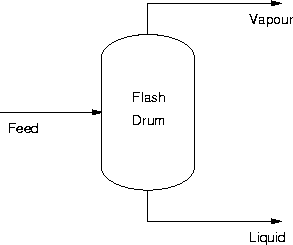
As you can see in this example there are:
- No. of Streams = 3
- No. of Extra Phases (interfaces) = 1
- CDF = 3 - 1 = 2
Therefore it is possible to control two strategic variables in an
adiabatic flash eg feed flowrate and pressure, leaving the third stream
free to determine the inventory.
The third example of this technique is a total condenser.
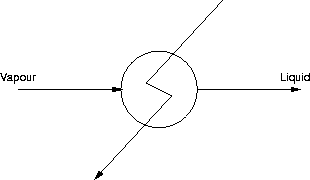
This time there are:
- No. of Streams = 3
- No. of Extra Phases (interfaces) = 1
- CDF = 3 - 1 = 2
Again it is possible to control two strategic variables.
A more complex unit that we can look at is a countercurrent cascade.
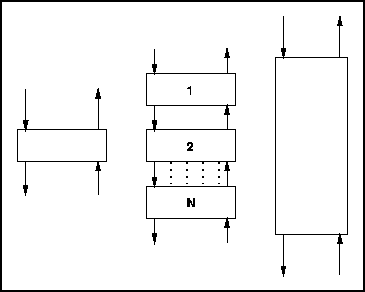
Figure 3 - Countercurrent Cascade
First consider a single countercurrent equilibrium stage:
- No. of Streams = 4
- No. of Extra Phases (interfaces) = 1
- CDF = 4 - 1 = 3
A stack of N such units built into a cascade will have 2N +
2 streams, N two phase elements and thus N + 2
apparant degrees of freedom. These would never all be used in practise
however, but it is, in principle, possible to maintain each stage at a
different pressure by some valve-like arrangement between the trays.
If there is a fixed rather than adjustable restriction between trays
then a potential degree of freedom is lost for each of N-1 vapour
interstreams, so the practical degrees of freedom for this device is
[N + 2] - [N - 1] = 3.
This is the same number of degrees of freedom as would be calculated by
simplistically counting the streams to the complete cascade, 4,
and subtracting 1 for the presence of two phases in the device,
again giving 3 Control Degrees of Freedom
Determination of the total degrees of freedom for a complete process is
now trivial. Connected units sharing a stream lose one degree of
freedom from the sum of those for the individual units. Thus the stream
related degrees of freedom is simply equal to the total number of
material and energy streams in the process.
The degrees of freedom for the complete process may be determined by
either of two equivalent procedures.
- Using the approach above determine the degrees of freedom for each
unit. Sum these and then subtract the number of shared streams to
obtain the final count.
- Count all the streams in the process. Separately count the total
number of extra phases i.e. add up all occurances of phases
greater than one in all units.
Method 2 is shown in the example below. All streams represent potential
degrees of freedom and possible adjustable variables, but beside each
unit is written the number lost as a result of the presence of multiple
phases in the unit.
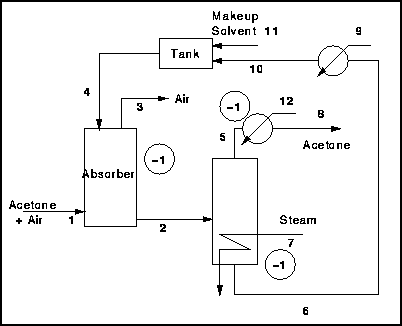
Figure 4 - Absorption Process with Solvent Recovery Flash Separator
- Total Streams = 12
- Extra Phases = 3
- Total D.O.F = 9
In the preceeding analysis it has been assumed that
- Material balances will always be somehow balanced.
- An adjustment which affects only inventory is not as such a
control degree of freedom.
An alternative view of 2 is that inventory control should be regarded as
a taking up a degree of freedom. Under this assumption the number of
degrees of freedom will always equal the number the material and energy
streams. Thus for the countercurrent cascade there would be 4 degrees
of freedom rather than 3, the additional one being the inventory of the
liquid phase. This alternative approach does have the advantage of
identifying all the potential control loops, explicit or implicit, which
will exist in a process.
It is quite straightforward to identify when and where inventory
regulation should be provided. Two trivial but useful rules are used in
this decision.
- If n streams join together in a process or part of a process over
which mass must be conserved - normally any process - then the
flow of only [n-1] these may be set by control loops other than one
regulating inventory within the process or part process.
- In any unit containing P phases, P control loops -
explicit or implicit - must be provided to maintain P
inventories, which may include combinations of phases. This number
allows for a loop to regulate total inventory.
Finally there appears to be three circumstances in which inventory
affects strategic process variables and thus should be counted as a
control degree of freedom.
- In the case of holdup dependent reaction conversion.
- In situations where inventory determines pressure.
- The construction of certain types of equipment means that a change
in holdup will affect performance e.g. by changing the surface area in a
flooded condenser.
It should be noted that where inventory affects several strategic
variables there will still only be one degree of freedom.
Return
to Start of Part 2: Further Developments






