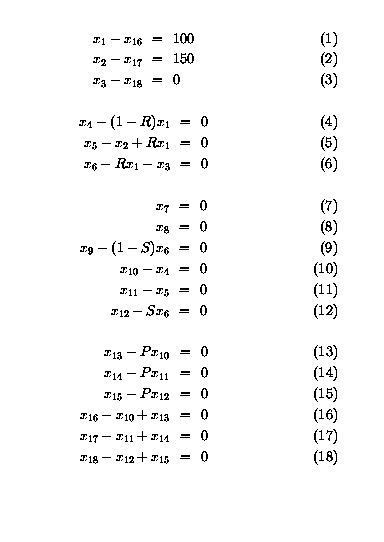Section 3.7.2: Process Case Study
You should first look at some revision material on material balances in
section 1.2.1.
We describe here a simple recycle chemical process
which will be used in a case study to illustrate a number
of aspects of steady-state modelling and
algebraic equation solving.
Process Description
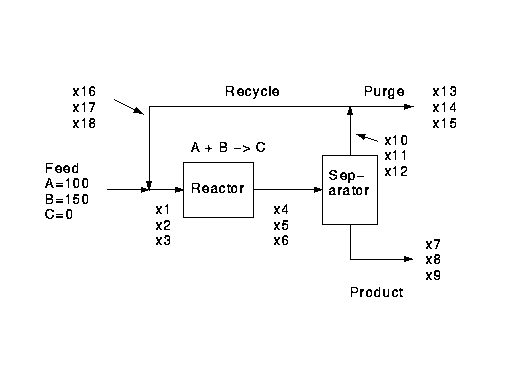
The process is to make product C from reactants A and B by the reaction
A + B -> C
It is fed with a mixture of A and excess B containing none of
the product material C.
The conversion in the reactor is R based on A.
Following the reactor all unreacted A and B are recovered
in the overheads of a distillation column and recycled.
The product C is not fully separated, a fraction S of that fed
to the separator remains in the overheads.
To avoid the buildup of unreacted B, a fraction
P of the overhead stream is removed as a purge.
Simple Process Model
The parameters of the model are:
- The fractional conversion R in the reactor, typically 0.8
- The fractional recovery S of C in the column overheads,
typically 0.1
- The fraction P of the overhead stream which is purged,
typically 0.05
18 variables x1 to x18 can be
associated with
the 3 molar flows of each of the species A, B and C in the 6
unknown process streams as shown in the flowsheet above.
Here x1 is the flowrate of A entering the reactor,
x2 that of B, and x3 that of C.
Similarly x4 is the flowrate of A out of the reactor, and
so forth.
The operations are all described by linear equations, as discussed
in section 1.2.1.
hence the whole process amy be described by a set of 18 linear equations as shown below.
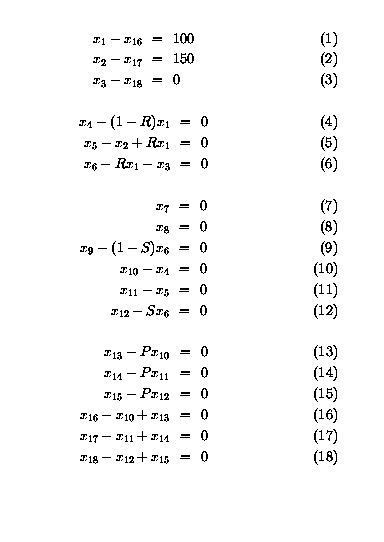
Return to Case Study Index
Return to Section 3 Index